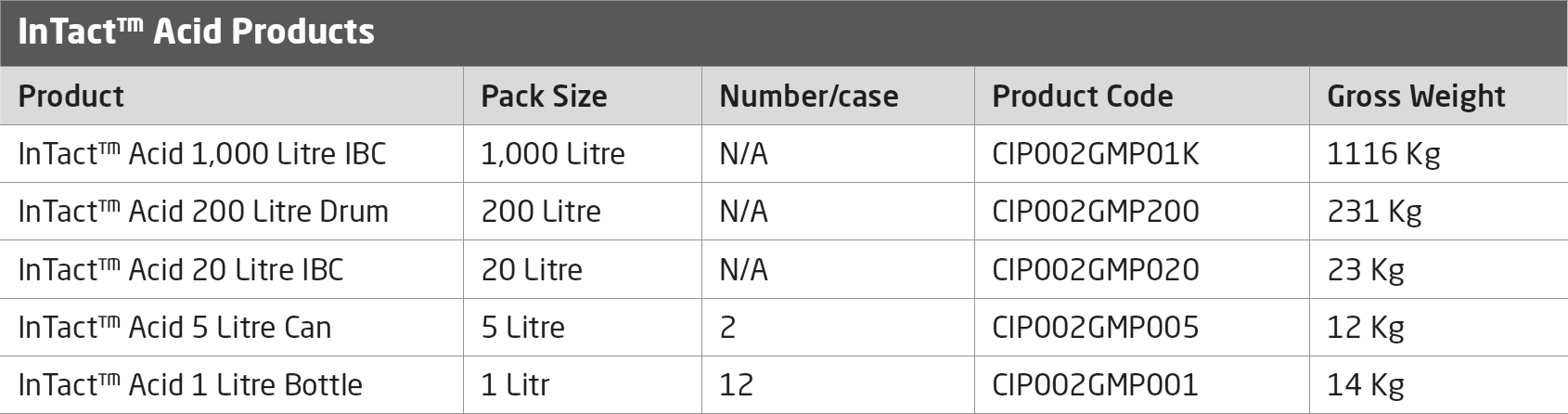
Overview
Redditch Medical's InTact™ products are so named because TACT describes the key parameters involved in cleaning optimisation: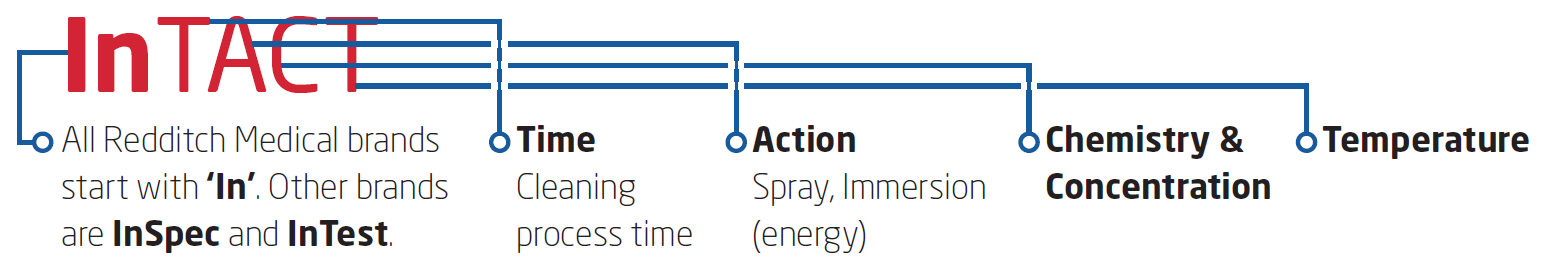
Description and Benefits
InTact™ Acid is a unique low foaming, low residue, Clean-in-Place (CIP) and Clean-out of-Place (COP) concentrated acid detergent formulation based on Phosphoric acid, biodegradable surfactants and chelants. InTact™ Acid is designed exclusively for use in GMP Pharmaceutical and Medical Device Industries and validation is supported with:
- Pre-qualified analytical test methods - TOC and ICP-MS
- Toxicology & formulation profile – releasable to end-users under a non-disclosure agreement
Use and Application
InTact™ Acid is suitable for use in all Clean-in-Place & Clean-out of-Place component washers and ultrasonic bath systems and due to the use of Phosphoric acid in the formulation may be used to specifically to de-rouge the stainless steel of components or the washer itself. Clean-in-Place monitoring systems can also make use of the data held on InTact™ Acid viscosity and conductivity at frequently used dilution rates.
If additional surfactancy is required InTact™ Acid is full compatible with InTact™ Neutral Booster – refer to Technical Datasheet/InTact Neutral Booster Rev 001.
Manufacture
InTact™ Acid is manufactured in an ISO 9001 and ISO 14001 registered facility with full batch traceability and complies with (EC) No. 648/2004 on detergent biodegradability.
InTact™ Acid is released with a certificate of analysis a two year expiration date for the date of manufacture. Safety data sheets (SDS) are available online or upon request.
Safe Handling
Always wear gloves and goggles or face protection, protective clothing. Always read the label and MSDS before use.
Conditions for Storage
Store in the tightly-closed, original container in a dry, cool and well-ventilated place. Keep upright to prevent leaking. Keep only in the original container. Corrosive storage. Additional test work supports that InTact™ Acid is unaffected by low temperatures –see freeze/thaw study – ref Freeze/thaw Study Rev 001.

Additional Supporting Services - InTact Optimisation Studies
Redditch Medical offer a laboratory-based service in which stainless steel or glass coupons representing an end-users actual drug & excipient soiling are tested in our laboratory to determine which cleaning agent should be used and at what concentration, time & temperature whilst mimicking cleaning process conditions.
The results from these studies provide both the rationale behind product selection and an optimised starting point to move to a pilot or pre-qualification trial with high assurance of success.
The service can also assist in reducing current cleaning times & rinsing volumes.
Processed coupons are reviewed and examined via several criteria to rapid establish how effective the cleaning & rinsing process has been under each set of TACT conditions. Criteria are:
- Visual
- Odour
- Water break free test ASTM Method F 22-65
- ATP rapid detection (for protein based soils)